When you switch to a generic drug, you might assume it’s exactly the same as the brand-name version. And technically, it is - same active ingredient, same dose, same FDA approval. But here’s the catch: how your body reacts to that drug might not be the same as someone else’s, even if you’re both taking the exact same pill. That difference? It’s often written in your genes - and sometimes, in your family’s medical history.
Why Two People React Differently to the Same Generic Drug
Imagine two people with high blood pressure. Both are prescribed the same generic version of lisinopril. One feels fine. The other gets dizzy, nauseous, and ends up in the ER. What happened? It’s not a manufacturing error. It’s not a mistake in dosing. It’s genetics. About 20% to 95% of how a person responds to a drug comes down to their DNA, according to the American Academy of Family Physicians. That’s huge. It means two people could have the same diagnosis, same prescription, same pharmacy - and still have wildly different outcomes. This isn’t new science. Back in the 1950s, doctors noticed some patients went into prolonged muscle paralysis after receiving a muscle relaxant called succinylcholine. Turns out, those patients had a rare genetic variation in an enzyme that breaks down the drug. Their bodies just couldn’t process it fast enough. Today, we call this field pharmacogenetics - the study of how genes affect drug response.Your Genes Are the Real Prescription
Your body uses enzymes to break down medications. The most important ones? The cytochrome P450 family. Among them, CYP2D6 is the superstar - and the troublemaker. It handles about 25% of all prescription drugs, including common ones like antidepressants (sertraline, paroxetine), beta-blockers (metoprolol), and painkillers (codeine). Here’s the problem: CYP2D6 has over 80 known genetic variants. People fall into categories:- Ultra-rapid metabolizers: Their bodies break down drugs too fast. The medication never builds up enough to work. A person on codeine might feel no pain relief because their body turns it into morphine too quickly.
- Normal metabolizers: The majority. The drug works as expected.
- Intermediate metabolizers: Slower breakdown. Might need a lower dose.
- Poor metabolizers: Their bodies barely process the drug. It builds up. Toxic levels. Serotonin syndrome. Liver damage. Hospitalization.
One study found that 7% of Caucasians and 10% of East Asians are poor metabolizers of CYP2D6. That means for a simple generic antidepressant, one in ten people could be at risk - and no one would know unless they got tested.
Family History Isn’t Just About Disease - It’s About Drug Reactions
You might think, “I’ll just ask my doctor if my family had bad reactions to meds.” And you should. But here’s why that’s not enough. If your mother had a severe rash on amoxicillin, that might be an allergy. But if your father passed out after taking warfarin - a blood thinner - that’s likely a genetic issue. Warfarin is metabolized by CYP2C9 and VKORC1. People with certain variants need 30% less of the drug. A family history of unexplained bleeding or clotting? That’s a red flag. Same with thiopurines - drugs used for leukemia and autoimmune diseases. If a relative had a life-threatening drop in white blood cells after taking azathioprine, they probably had a TPMT gene variant. That’s hereditary. And if you inherit it, your risk is just as high. Family history doesn’t just tell you what diseases you might get. It tells you how your body will handle the drugs meant to treat them.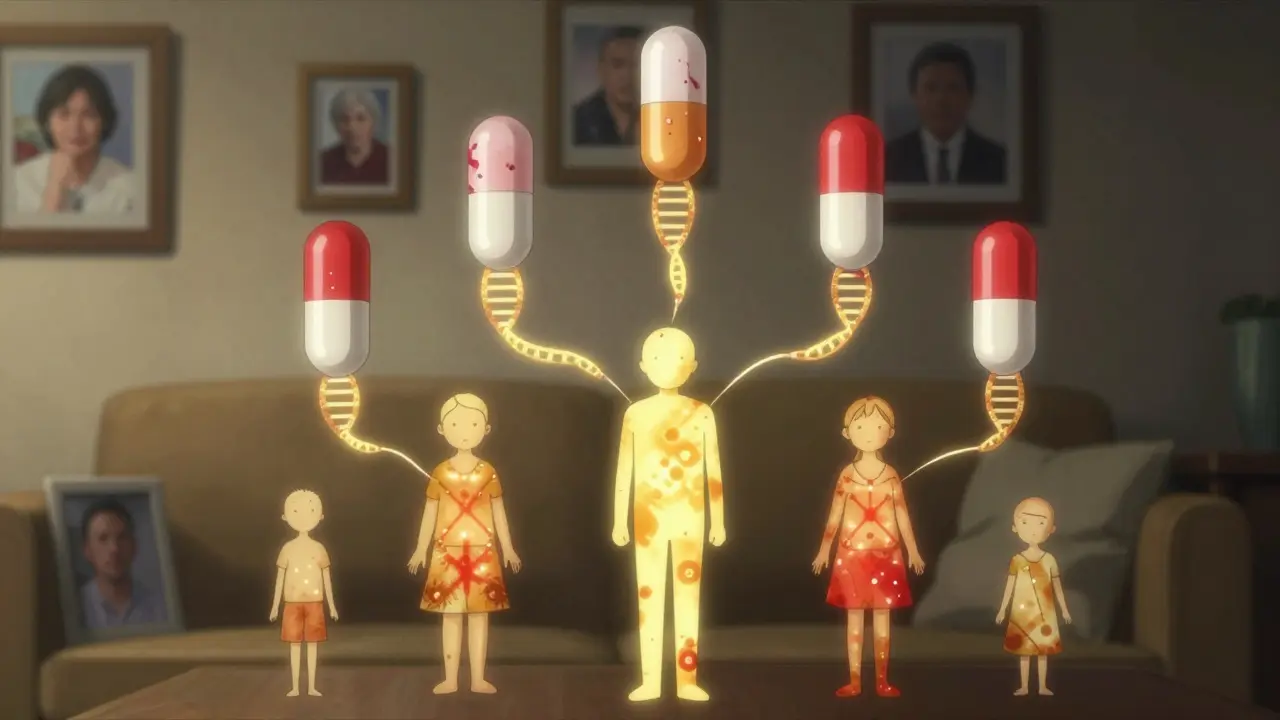
Why Generics Can Be Riskier Than You Think
Switching to generics makes sense. They’re cheaper. They’re just as effective - for most people. But if your body processes drugs differently? That “just as effective” doesn’t apply. A 2023 Mayo Clinic study tracked 10,000 patients who had preemptive genetic testing before starting medications. Over 40% had at least one high-risk gene-drug interaction. In 67% of those cases, doctors changed the drug or dose. The result? A 34% drop in adverse events. Now picture this: You’re on a generic version of fluoxetine. You’ve been fine for six months. Then you switch pharmacies. The new generic has a different inactive ingredient - not the active drug, but the filler. That’s allowed. But if you’re a CYP2D6 poor metabolizer, even a tiny change in absorption can push your blood levels into toxic range. No one warned you. No one tested you. And now you’re in the ER with serotonin syndrome. Generics aren’t the problem. The problem is assuming everyone responds the same way.Population Differences Matter - Even in the UK
Genetic variants aren’t spread evenly. The CYP2C19 gene, which affects how you metabolize proton pump inhibitors (like omeprazole), shows clear differences. About 15-20% of Asians are poor metabolizers. Only 2-5% of Europeans are. That means a standard dose of omeprazole might work great for your neighbor but leave you with uncontrolled acid reflux. Warfarin dosing varies too. African ancestry populations often need higher doses due to genetic differences in CYP2C9 and VKORC1. But if your doctor just uses a one-size-fits-all chart based on age and weight? You’re at risk of clotting or bleeding. This isn’t just a US issue. The UK’s NHS uses the same generic drugs. And if your GP doesn’t know your genetic background - or worse, assumes you’re “typical” because you’re white British - you could be getting the wrong dose.
What’s Being Done - And What You Can Do Now
The FDA now lists over 300 drugs with pharmacogenetic information on their labels. Warfarin, clopidogrel, abacavir, and several cancer drugs all come with genetic warnings. The Clinical Pharmacogenetics Implementation Consortium (CPIC) has published 24 guidelines to help doctors use this data. But adoption is slow. Only 32% of community hospitals in the UK and US have pharmacogenomic programs. Most GPs don’t know how to interpret a CYP2D6 result. And testing? It’s not routine. Here’s what you can do:- Ask your doctor: “Have you ever seen someone in our family have a bad reaction to a medication?”
- Check your family history: Did a relative have a severe rash, unexplained bleeding, or sudden side effects after starting a new drug?
- Consider a test: Tests like Color Genomics or OneOme cost around £200-£400. They look at 10-15 key genes. If you’re on multiple medications, it’s worth it.
- Bring results to your pharmacist: Pharmacists are trained to read these reports. They can flag dangerous combinations.
One patient on Reddit shared that after a DPYD gene test showed she was a slow metabolizer, her oncologist cut her 5-fluorouracil dose in half. She finished chemotherapy without a single hospital visit. That test cost £250. It saved her life.
The Future Is Personal - Not Generic
The future of medicine isn’t “one size fits all.” It’s “one size fits you.” The All of Us Research Program in the US is already returning genetic results to over a million people. The UK is starting pilot programs too. By 2025, most academic hospitals plan to offer preemptive genetic testing. But until then, you’re your own best advocate. If you’re switching to a generic drug - especially if you’re on antidepressants, blood thinners, seizure meds, or chemotherapy - don’t assume it’s safe just because it’s cheap. Ask about your genes. Ask about your family. And if your doctor shrugs? Get a second opinion. Your genes didn’t choose your medication. But they’re the ones who will decide if it works - or if it harms you.Can family history predict how I’ll respond to generic drugs?
Yes - but not perfectly. If a close relative had a severe reaction to a specific drug - like unexplained bleeding on warfarin, a dangerous rash on carbamazepine, or extreme nausea on SSRIs - that’s a strong signal you might share the same genetic variant. Family history can point you toward testing, but it can’t replace it. Some gene variants are silent until you take the drug. Only genetic testing can confirm your risk.
Are generic drugs less safe than brand-name drugs because of genetics?
No. Generic drugs are chemically identical to brand-name versions. The problem isn’t the drug - it’s your body’s ability to process it. A poor metabolizer might have a bad reaction to brand-name sertraline just as easily as to the generic. The risk comes from assuming everyone reacts the same. That’s why genetic testing matters, not the drug label.
Is pharmacogenetic testing covered by the NHS?
Currently, the NHS does not routinely offer pharmacogenetic testing outside of very specific cases - like testing for HLA-B*15:02 before carbamazepine in epilepsy patients. For most drugs, including antidepressants or blood thinners, testing is not funded. You can get it privately through companies like Color Genomics or OneOme. Some NHS hospitals are running pilot programs, but widespread access isn’t expected until at least 2027.
Which drugs have the strongest genetic links?
The strongest evidence exists for:
- Warfarin: CYP2C9 and VKORC1 genes determine safe dose.
- Clopidogrel: CYP2C19 poor metabolizers don’t convert it to active form - higher risk of heart attack.
- Abacavir: HLA-B*57:01 carriers risk a life-threatening hypersensitivity reaction.
- Thiopurines (azathioprine): TPMT variants cause severe bone marrow suppression.
- 5-Fluorouracil: DPYD variants cause toxic buildup - can be fatal.
- SSRIs and codeine: CYP2D6 variants cause treatment failure or overdose.
What if my doctor says genetic testing isn’t necessary?
It’s your right to ask. If you’re taking multiple medications, have a family history of bad reactions, or experienced side effects yourself, push for testing. You can request a referral to a clinical pharmacogenetics service. Many academic hospitals have them. If your GP refuses, ask for a second opinion from a pharmacist with pharmacogenetics training. The evidence is solid - and your safety is worth it.






Erin Pinheiro
February 24, 2026 AT 18:51Okay but like… have you ever tried to get a doctor to take you seriously when you say your mom died from a "simple" generic antidepressant? No? Yeah. Me neither. They just nod and hand you another script. Like, it’s 2025 and we’re still playing Russian roulette with our meds. I swear, if I hear "it’s just a placebo effect" one more time…
Michael FItzpatrick
February 25, 2026 AT 06:05Let me tell you something that ain’t in the FDA pamphlets: your body doesn’t care about brand names or generic labels. It cares about enzymes. And if your CYP2D6 is throwing a tantrum, no amount of corporate marketing is gonna fix that. I’ve seen people go from "I’m fine" to "why is my tongue numb?" in 48 hours because they switched pharmacies. This isn’t sci-fi. It’s Tuesday in America.
Ashley Johnson
February 26, 2026 AT 19:58Big Pharma knows this. They don’t test for genetics because if they did, they’d have to pay for it. And if they paid for it, they’d have to lower prices. And if they lowered prices, the stock would crash. So they let you bleed out in your living room while they sell you the same pill in a different color. Wake up.
tia novialiswati
February 27, 2026 AT 09:03Hey, I just got my gene test back and wow-turns out I’m a CYP2D6 poor metabolizer. My doc didn’t even blink. But my pharmacist? She printed out a whole chart. We changed my meds. I haven’t felt dizzy in 3 months. 🙌 You guys are not alone. Ask. Test. Advocate. It’s worth it.
Lillian Knezek
February 28, 2026 AT 03:00Did you know the government is hiding this? They’ve been testing people since the 90s and burying the results. Your pharmacy is linked to a database that flags "high-risk" patients. But they don’t tell you. Why? Because if you knew, you’d stop buying generics. And they need you hooked. 💀
Maranda Najar
March 1, 2026 AT 20:52Oh. My. GOD. I just read this and I’m sobbing. My sister. She was 27. Took a generic sertraline. Said she felt "off." Three days later, she was in a coma. They said it was "unexplained." But her aunt had the same thing. And her grandfather? Died after a codeine pill. We didn’t connect the dots. We didn’t know. Now I’m begging everyone I know to get tested. Please. Don’t wait until it’s too late.
Sanjaykumar Rabari
March 1, 2026 AT 21:11India also has same problem. Many people take generic medicines. No testing. No warning. My uncle died after taking generic clopidogrel. Doctors said "it was heart attack." But his brother had same thing. We never knew about CYP2C19. Now I tell everyone. Test before you take.
Kenzie Goode
March 2, 2026 AT 13:54This is one of those posts that makes you rethink everything you thought you knew about medicine. I used to think generics were just cheaper versions of the same thing. Now I realize they’re the same chemical-but our bodies are different languages. And no one taught us how to read them.
Dominic Punch
March 4, 2026 AT 11:51As a pharmacist in Manchester, I see this every week. A woman comes in with a rash after switching from brand to generic fluoxetine. She’s terrified. I check her meds, ask about family history-turns out her mum had serotonin syndrome on the same thing. We pull the genetic report. Poor metabolizer. Changed her drug. No more ER visits. This isn’t theoretical. It’s my job. And we’re still not doing enough.
Lou Suito
March 4, 2026 AT 12:30Nick Hamby
March 4, 2026 AT 21:47There’s a profound philosophical shift here: medicine is no longer about treating disease-it’s about aligning with biology. The assumption that all human bodies are interchangeable is not just outdated; it’s lethal. We’ve built systems on homogeneity, but biology is a mosaic. To treat a person as a statistical average is to ignore their uniqueness. The future isn’t just personalized-it’s individualized. And if we don’t evolve, we’ll keep burying people who took the right pill… for someone else.
kirti juneja
March 5, 2026 AT 12:24My cousin in Delhi took generic warfarin and almost died. No one told her about VKORC1. We found out only after she went to a private clinic. Now she pays out of pocket for testing. I told my whole family. If you take meds? Get tested. £200 is cheaper than a funeral. 💔
Haley Gumm
March 7, 2026 AT 01:12Let’s be real: if your family has a history of bad reactions, you’re basically a walking liability. And if you’re on more than three meds? Congrats-you’re a pharmacological minefield. Someone should’ve told you this before you started. But here we are. Now you know. Go get tested. Or don’t. But don’t say you weren’t warned.